Influential medical magazine 'The Lancet' gives Kazakh- produced COVID vaccine QazCovid-in® top marks
 27 Августа 2021
27 Августа 2021
 1143
1143
 Медицина,
Наука
Медицина,
Наука
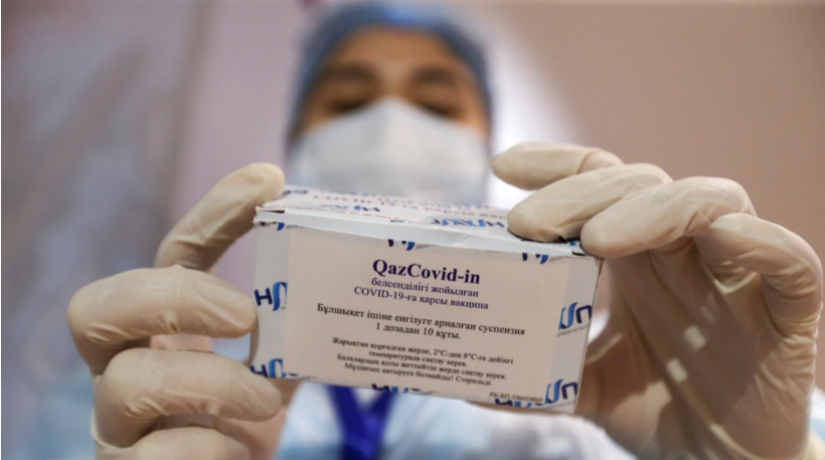 Фото:EU Reporter
Фото:EU Reporter
Almaty. August 27. Information Center - The influential UK medical magazine The Lancet has published an expert review which gives a positive report on the effectiveness of the Kazakh produced Covid vaccine QazCovid-in®, EU Reporter reports.
In its EClinicalMedicine online review published in the middle of August, it concluded that: “QazCovid-in® is an inactivated aluminium adjuvanted whole-virion vaccine against COVID-19. Our phase 1 and 2 clinical trials have proved the QazCovid-in® vaccine to be safe and well-tolerated by adults aged from 18 to 70 years, who receive one or two doses.
“The reported local and systemic AEs, associated with intramuscular administration of the inactivated vaccine, are predominantly mild and classified as foreseeable.
"Between the vaccine and placebo groups, no statistically significant difference has been shown in the level of total serum IgE antibodies which are known as specific triggers of allergic reactions .
"A good safety profile is a big advantage of the QazCovid-in® vaccine which does not induce acute allergic reactions.”
Between 19-23 September 2020, 104 potential participants aged 18–50 years were screened, of whom 44 were enrolled into the phase 1 clinical trial and randomly assigned to receive the QazCovid-in® vaccine or placebo.
The report states: “Participants were recruited through ethics-approved advertising on the study site's website. Eligible participants were male or female adults aged 18 years and older who were healthy at enrolment based on their medical history, vital signs, and physical examination. Participants were screened to meet the inclusion criteria."
Subjects were enrolled in the trials if they had no history of COVID-19, their serology tests for IgM and IgG antibodies to SARS-CoV-2 were negative, and if they had no close contact with individuals suspected of being infected with SARS-CoV-2 14 days before the enrolment.
Female participants of childbearing potential had to agree to use reliable forms of contraception throughout the whole study period. Volunteers were screened for the absence of chronic viral infections, such as human immunodeficiency virus (HIV), hepatitis B (HBV) and hepatitis C (HCV) viruses.
Routine biochemical blood test, hematology test panel, electrocardiogram (ECG), and pregnancy test for women were also performed. Main exclusion criteria included allergic history, drug intolerance including hypersensitivity to any vaccine component, an axillary temperature of more than 37·0 °C, abnormalities in laboratory tests, and positive urine pregnancy test for women.
Subjects with any mental disease or serious chronic illness were also excluded. A detailed list of the inclusion and exclusion criteria can be found in the Supplement. T
The trials were approved by the National Regulatory Authority and the ethics committee of the National Scientific Centre for Phthisiopulmonology of the Republic of Kazakhstan (No. KZ78VMX00000211) and conducted in compliance with the International Council for Harmonization Good Clinical Practice guidelines.
An investigator or a staff member explained the investigational nature and the purpose of the study to a subject in sufficient detail to allow the subject to make an informed decision about participating. Written informed consent was obtained from the study subjects before enrolment and before any study procedure, including screening. The trials were registered with ClinicalTrials.gov NCT04530357.
While the sample size was not based on any statistical hypothesis, it was adjusted to adequately assess the vaccine's safety in each study group. Qualitative characteristics of a study group were presented as percentages; quantitative parameters were presented as median with interquartile range (IQR) and min-max range.
The safety analysis set included all randomised participants who received at least one dose of the study vaccine or placebo. Descriptive summary data were provided as numbers and per group percentages and included the participants who reported at least one solicited local reaction or systemic adverse event, any unsolicited adverse events, serious adverse events, or adverse events of special interest after the first or second dose.
The attributable risk was calculated for particular groups and presented with 95% CI calculated by the Newcombe-Wilson method with continuity correction
The work was funded by the Science Committee of the Ministry of Education and Science of the Republic of Kazakhstan. The sponsor had no role in the design of the study, as well as in the collection, analyses, or interpretation of data, or in the writing of the manuscript, and in the decision to publish the results.
To date, 6,183,445 people have received their first dose of the COVID-19 vaccine. 4,819,587 citizens of the republic have been fully immunized.
The population of Kazakhstan is 19 million.
ru Человек-эпоха: Куаныш Ажмагамбетов и его вклад в развитие промышленности Жетісу
ru Займы под 120% годовых: дело о незаконном кредитовании передано в
ru Выставка тепличных технологий Grow Expo Astana 2026 пройдет 8-10 апреля
ru Жаркентская мечеть включена в предварительный список Всемирного наследия ЮНЕСКО
ru В Минтруда обсудили новые механизмы трудоустройства лиц с инвалидностью
ru Схема обмана студентов через соцсети раскрыта в Шымкенте
ru В Акорде состоялась церемония встречи Президента Сербии
ru Мобильный ЦОН: сколько услуг казахстанцы получают через приложение
ru Подростки с особыми потребностями осваивают профессию кондитера в Карагандинской области
ru КНБ ликвидировал канал контрабанды наркотиков
ru Охлажденное мясо птицы можно будет хранить в 3 раза дольше
ru Солдат Национальной гвардии разработал цифровой портал для военнослужащих
ru Учащиеся 0-9 классов второй смены переведены на дистанционный формат обучения
ru Пенсионерка едва не лишилась более 2 млн тенге в Павлодаре
ru Строительство нового завода Coca-Cola Içecek в Актобе планируется начать в
ru Один из крупнейших бизнесменов Польши построит фармзавод в Шымкенте
ru Стартует прием заявок на премию Президента Республики Казахстан «Алтын Сапа»
ru Президент Сербии Александр Вучич посетит Астану 26-27 февраля
ru Глава государства направил поздравительную телеграмму Эмиру Кувейта
ru МТСЗН РК опровергло фейк о ежегодном отпуске
ru В Алматы открылась юбилейная выставка о мастерах кино
ru В Астане вынесен приговор интернет-мошеннице
ru Олжас Бектенов о выделении 1 трлн тенге на посевную кампанию
ru Олжас Бектенов поручил упростить и оцифровать процедуру льготного кредитного финансирования
ru В Казахстане продолжается подготовка к паводковому периоду
ru В Алматы продолжают расширять фонд студенческих общежитий
ru Почти 3000 га сельхозземель возвращены в госсобственность в Туркестанской области
ru Марихуана в особо крупном размере изъята в ВКО
ru Олжас Бектенов провел заседание Совета директоров «Самрук-Қазына»
ru Из Турции экстрадирован подозреваемый в серийном мошенничестве

 +77772555856
+77772555856
 Написать нам
Написать нам












